The role of structural disorder in the rewiring of protein interactions through evolution
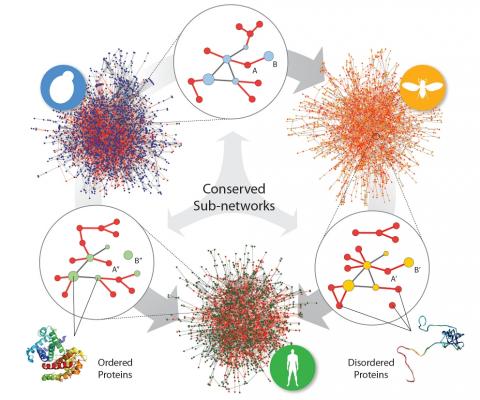
Mosca M, Pache RA, Aloy P, Structurally disordered regions play a key role in protein-protein interaction networks and the evolution of highly connected proteins, enabling the molecular mechanisms for multiple binding. However, the role of protein disorder in the evolution of interaction networks has been only investigated through the analysis of individual proteins, being impossible to distinguish its specific impact in the (re) shaping of their interaction environments. Now, the availability of large interactomes for several model organisms permits to explore the role of disorder in protein interaction networks not only at the level of the interacting proteins, but of the interactions themselves. By comparing the interactomes of human, fly and yeast, we discovered that, despite being much more abundant, disordered interactions are significantly less conserved than their ordered counterparts. Furthermore, our analyses provide evidence that this happens not only because disordered proteins are less conserved, but also because they display a higher capacity to rewire their interaction neighborhood through evolution. Overall, our results support the hypothesis that conservation of disorder gives a clear evolutionary advantage, facilitating the change of interaction partners during evolution. Moreover, this mechanism is not exclusive of a few anecdotal cases but a global feature present in the interactome networks of entire organisms.
Mol Cell Proteomics,
2012
Pubmed: 22389433
Direct link: 10.1074/mcp.M111.014969
